1. Polycarbonates
Carbon dioxide, is a highly abundant, nontoxic, nonflammable, C1 feedstock which can be copolymerized with epoxides to generate polycarbonates. These materials have a wide range of potential applications including use in polyurethanes and specialty products. Our group studies highly active β-diiminate zinc ([BDI]ZnOAc) catalysts which have allowed us to access a plethora of different polycarbonates, including isotactic polymers via meso desymmeterization, and poly(limonene carbonate) which shows a unique example of stereocomplexation.
-
Ellis, W. C.; Jung, Y.; Mulzer, M.; Di Girolamo, R.; Lobkovsky, E. B.; Coates, G. W. Chem. Sci., 2014, 5, 4004-4011. doi: 10.1039/C4SC01686F
-
Auriemma, F.; De Rosa, C.; Di Caprio, M. R.; Di Girolamo, R.; Ellis, W. C.; Coates, G. W. Angew. Chem. Int. Ed., 2015, 54, 1215–1218. doi: 10.1002/anie.201410211
-
Vidavsky, Y.; Yang, S. J.; Abel, B. A.; Agami, I.; Diesendruck, C. E.; Coates, G. W.; Silberstein, M. N. J. Am. Chem. Soc., 2019, 141, 10060–10067. doi: 10.1021/jacs.9b04229
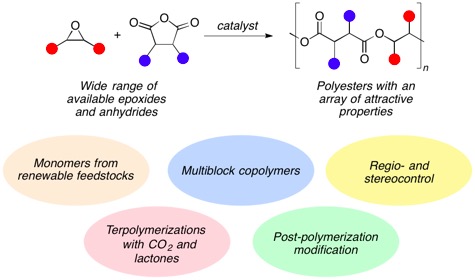
2. Polyesters
Our group has developed zinc, chromium, cobalt, and aluminum catalysts for the alternating copolymerization of epoxides and cyclic anhydrides. This chain-growth synthesis offers an attractive alternative to traditional step-growth methods for polyester synthesis. We can synthesize an array of diverse structures with widely varying properties, ranging from semi-crystalline stereocomplexed poly(propylene succinate), to completely amorphous high-Tg terpene based materials, to polyester-b-polycarbonate block copolymers via a one-pot polymerization.
-
Sanford, M. J.; Van Zee, N. J. and Coates, G. W. Chem. Sci., 2018, Advance Article. doi: 10.1039/C7SC03643D
-
De Hoe G. X.; Zumstein, M. T.; Tiegs, B. J.; Brutman, J. P.; McNeill, K.; Sander, M.; Coates, G. W.; Hillmyer, M. A. J. Am. Chem. Soc., 2018, 140, 963–973. doi: 10.1021/jacs.7b1017
-
Yu, X.; Jia, J.; Xu, S.; Lao, K.; Sanford, M. J.; Ramakrishnan, R. K.; Nazarenko, S. I.; Hoye, T. R.; Coates, G. W.; DiStasio Jr., R. A. Nature Communications, 2018, 9, 2880–2888. doi: 10.1038/s41467-018-05269-3
- Lidston, C. A.; Abel, B. A.; Coates, G. W. J. Am. Chem. Soc., 2020, 142, 20161−20169. doi: 10.1021/jacs.0c10014
3. Poly(hydroxyalkanoates)
Poly(hydroxyalkanoate)s (PHA)s are naturally-occurring biodegradable polyesters made by bacterial fermentation. Our goal is to develop an alternative synthetic route that consists of carbonylation of epoxides to beta-lactones, followed by ring-opening polymerization to yield PHAs. Recently, we reported a one-pot carbonylative polymerization method that makes poly(3-hydroxybutyrate) (PHB) from propylene oxide and carbon monoxide.
-
Lu, Y.; Swisher, J. H.; Meyer, T. Y.; Coates, G. W. J. Am. Chem. Soc., 2021, 143, 4119-4124. doi: 10.1021/jacs.1c00248